Obtained ISO 9001 certification for the first time in the Korean pharmaceutical industry.
Having the ability to produce a total of 200 finished pharmaceutical products and health functional foods.
CHOA SAFEASY Ampoule
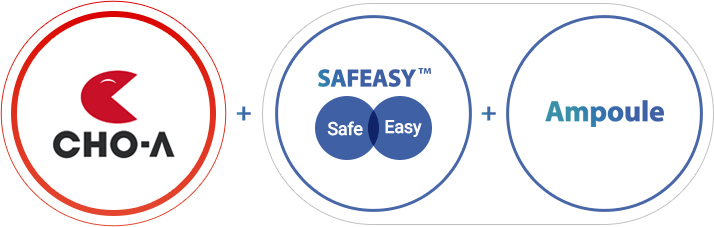

Uniqueness
- Unique design
- Ergonomic design
- Domestic and international patents (in 22 countries)
Safety
- Resistant design
- Enhanced durability and heat resistance
- Double protection against external impacts
Stability
- Block of impurity inflow
- Prevents destruction of ingredients
- Prevents deterioration of ingredients
Convenience
- Portable
- One-touch design
- Easy to use
Materials certified as USP Plastic Class VI exhibit excellent biocompatibility (safety) compared to various plastic materials.

What is USP Plastic Class VI certification?
The most stringent international standard for proving the biocompatibility of medical devices.
Dosage form Details
| Category | Dosage forms | ||
|---|---|---|---|
| Oral solids | Tablet | Tablet / Coated Tablet | |
| Capsule | Hard capsules | ||
| Granules | |||
| Chewable tablet | |||
| Oral liquids | Oral liquids Syrup (Liquid) |
Ampoule | Glass |
| SAFEASY | |||
| Liquid pouch (Sachet) | |||
| Bottle | |||
Major partners










Contact information
Consignment business Team | Sung-Won, Jung 
+82-2-6670-9355
Republic of Korea (Domestic)
Overseas Sales 1 Team | Chul-Min, Jung (Charles) 
+82-2-6670-9293
Central Asia, Middle east, Chinese-speaking countries
Overseas Sales 2 Team | Hyun-Kyu, Park (Leo) 
+82-2--6670-9294
South-East Asia, Africa, America
